IQVIA supports sites and participants through patient-centric trial experiences
Por um escritor misterioso
Descrição
In this interview, Eric Klaver, the DCT regulatory director for decentralized clinical trials at IQVIA, aiding in compliance of the DCT strategy and platform, shares how IQVIA supports investigator | With IQVIA’s clinical trial solutions and flexible site support strategies, decentralized trial elementss are being leveraged to enhance patient- centered care as well as support sites through hands- on enablement and implementation of DCT platforms these services.

Clinical Outcome Assessments Accelerator - IQVIA

Patient friction coefficient' can gauge a clinical trial's patient burden - STAT

CRO Trends: IQVIA Perspective
Everything You Wanted To Know About Decentralized Clinical Trials - TrialX

IQVIA on AI's potential to match patients to trials and improve trial diversity
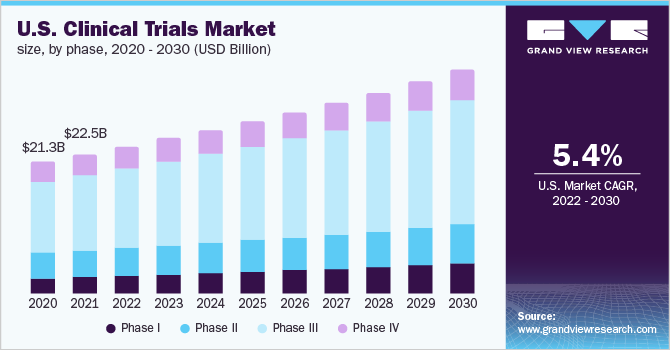
U.S. Clinical Trials Market Size & Share Analysis Report, 2030

IQVIA_HOLDINGS_INC.__________2023_Proxy_Statement

Patient Recruitment for Clinical Trials - IQVIA

Revolutionizing Drug Development: IQVIA's David Cameron on the Power of Innovative Trial Designs

Flexible Clinical Modeling: How Advanced Analytics and AI/ML Can Help Ensure Effective Patient-Centered Drug Development

Site Support: The Cornerstone of the Digitalization of Clinical Trials
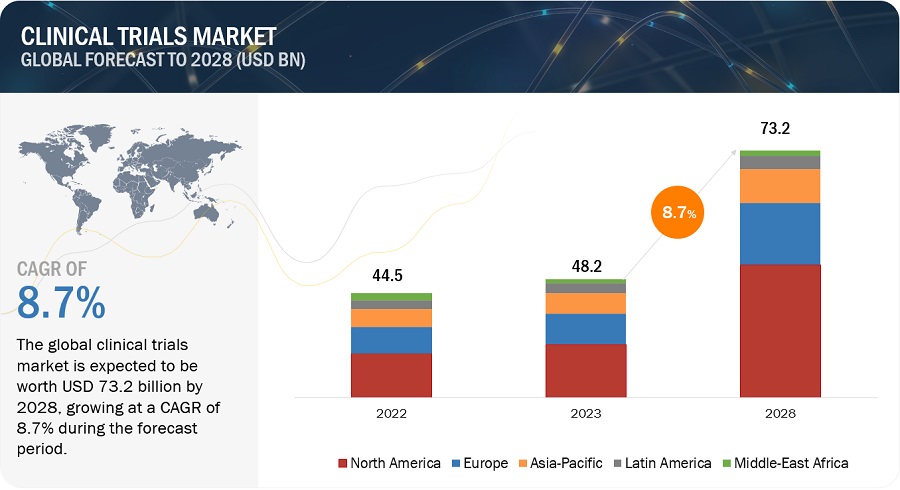
Clinical Trials Market Size, Share, Trends and Revenue Forecast [Latest]

Top 17 Clinical Research Organizations in 2022 [Ultimate Guide]
de
por adulto (o preço varia de acordo com o tamanho do grupo)


:max_bytes(150000):strip_icc()/logo-FiercePharma-7f82be3022eb4fb2872c3b8988d0d3e2.png)




